image
AI Democratizes Cancer Cures: Man Uses ChatGPT & AlphaFold to Save Dog with Custom mRNA Vaccine
The Garage Lab Miracle: A Dog’s Dramatic Recovery
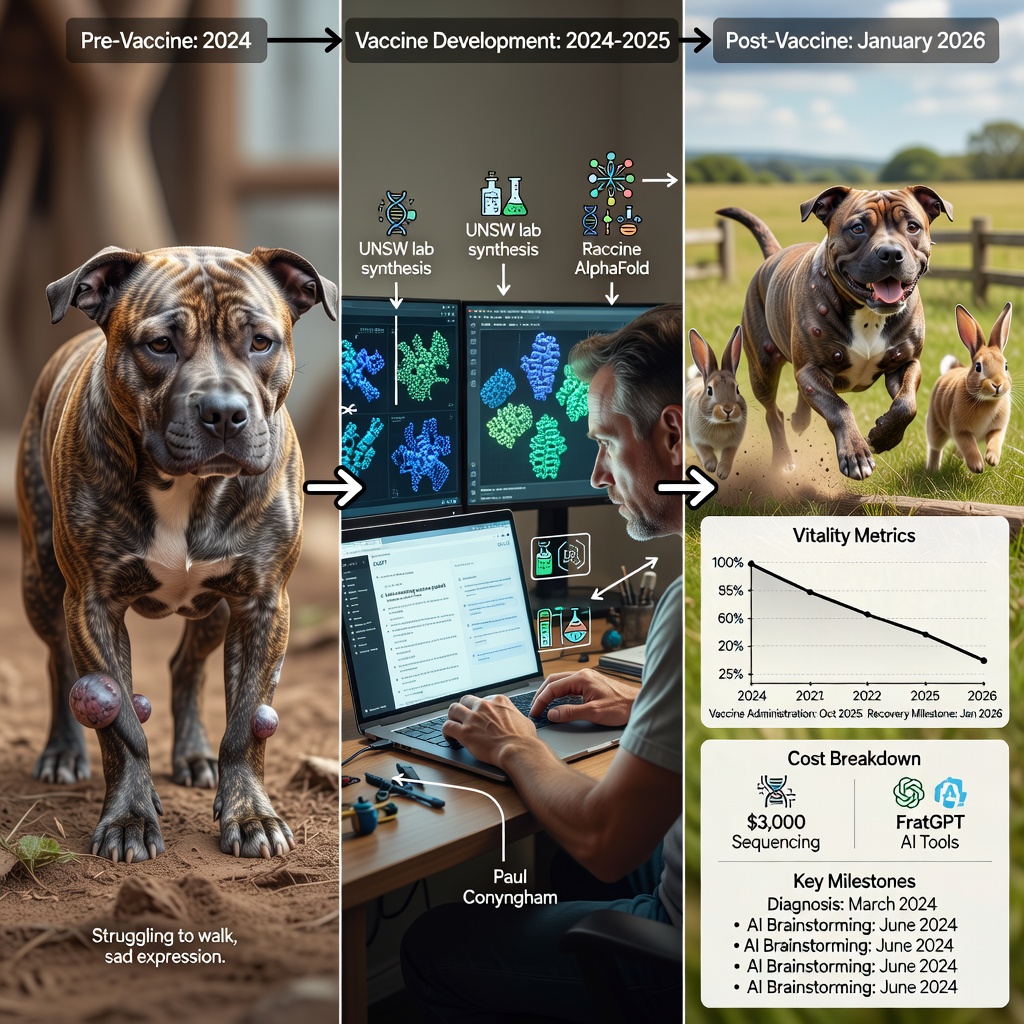
In the quiet suburbs of Sydney, Australia, a story unfolded in 2024 that reads like science fiction but is grounded in cutting-edge reality. Paul Conyngham, an AI consultant with no formal biology training, faced a heartbreaking crisis: his rescue dog Rosie, a spirited Staffordshire Bull Terrier crossed with a Shar Pei, was battling an aggressive mast cell tumor. Traditional veterinary interventions surgery and chemotherapy offered only temporary relief. The tumors grew to the size of tennis balls, robbing Rosie of her ability to walk comfortably. Vets delivered a grim prognosis: mere months to live.
Refusing to surrender, Conyngham approached Rosie’s cancer as a software glitch in need of debugging. He invested approximately $3,000 to sequence the tumor’s DNA at the University of New South Wales’ (UNSW) Ramaciotti Centre for Genomics. Armed with gigabytes of genetic data, he turned to publicly available AI tools: OpenAI’s ChatGPT for strategic brainstorming and Google DeepMind’s AlphaFold for protein modeling. ChatGPT helped map out a pipeline from identifying mutated proteins to hypothesizing vaccine targets while AlphaFold predicted their 3D structures in minutes, a task that once demanded years of lab work by PhD teams.
The result was a concise, half-page “blueprint” for a custom mRNA vaccine tailored precisely to Rosie’s tumor mutations. Conyngham collaborated with Prof. Páll Thordarson, director of UNSW’s RNA Institute, who oversaw the synthesis of the physical vaccine. In under two months, the vaccine was ready. Administered starting in late 2024, it triggered stunning results: the largest tumor on Rosie’s leg shrank by 75% within one month, with secondary tumors vanishing entirely. By January 2026, Rosie was bounding over fences and chasing rabbits, her vitality restored. Conyngham is now iterating a second-generation vaccine for a persistent mass, proving this wasn’t luck but reproducible science.
This marks the world’s first fully personalized mRNA cancer vaccine designed for a canine patient crafted not by a pharma giant, but by one determined individual with a laptop.
Echoes of History: Technology’s Relentless March to the Masses
Rosie’s revival isn’t an isolated anomaly; it’s the latest chapter in a familiar historical arc where elite technologies cascade into everyday hands, upending industries.
Consider computing: In the 1960s, IBM mainframes like the System/360 cost millions and filled entire rooms, accessible only to governments and corporations. By the 1980s, the IBM PC democratized processing power, and today, smartphones eclipse the Apollo Guidance Computer that landed humans on the Moon in 1969.

3D printing followed suit. Once confined to NASA’s labs for rocket parts or Boeing’s factories, it hit garages in the 2010s via affordable printers like the Prusa i3, enabling hobbyists to fabricate prosthetics or custom tools.
Laser cutters and CNC machines trace a similar path from industrial behemoths in the 1970s to desktop versions under $500 today, fueling maker movements and drone DIYers.
Medicine has lagged, gated by regulatory fortresses and billion-dollar R&D costs. Drug development averages $1-2 billion and 10-15 years, per Tufts Center for the Study of Drug Development data, prioritizing high-volume “blockbusters” over rare diseases or personalized therapies. Orphan cancers, affecting small patient pools, often languish deemed “unprofitable.” Conyngham’s feat shatters this: for $3,000 plus AI (free or low-cost), he bypassed the pipeline, echoing how the Homebrew Computer Club in 1970s Silicon Valley birthed the PC revolution.
AlphaFold’s Nobel Legacy: The Protein Puzzle Solved
Central to this triumph is AlphaFold, DeepMind’s AI that cracked the 50-year “protein folding problem” predicting how amino acid chains fold into functional 3D shapes, crucial for drug design. In 2024, DeepMind’s Demis Hassabis and John Jumper shared the Nobel Prize in Chemistry for this breakthrough, hailed by the Royal Swedish Academy as transformative for biology.
Paul didn’t need a supercomputer or doctorate. AlphaFold’s open database (over 200 million structures) let him model Rosie’s mutated mast cell proteins instantly. ChatGPT orchestrated the workflow: analyzing sequencing data, selecting neoantigens (tumor-specific proteins), and outlining mRNA sequences to train Rosie’s immune system against them. This mirrors human trials, like Moderna-Merck’s mRNA-4157 for melanoma, but Conyngham executed it solo, faster, and for a non-human patient.
From a technical perspective, this leverages mRNA’s post-COVID maturity platforms honed by Pfizer-BioNTech vaccines now enable rapid customization. UNSW’s synthesis proves academic labs can scale “wild ideas” without corporate overhead.
Perspectives: Empowerment vs. Peril in the AI-Medicine Clash
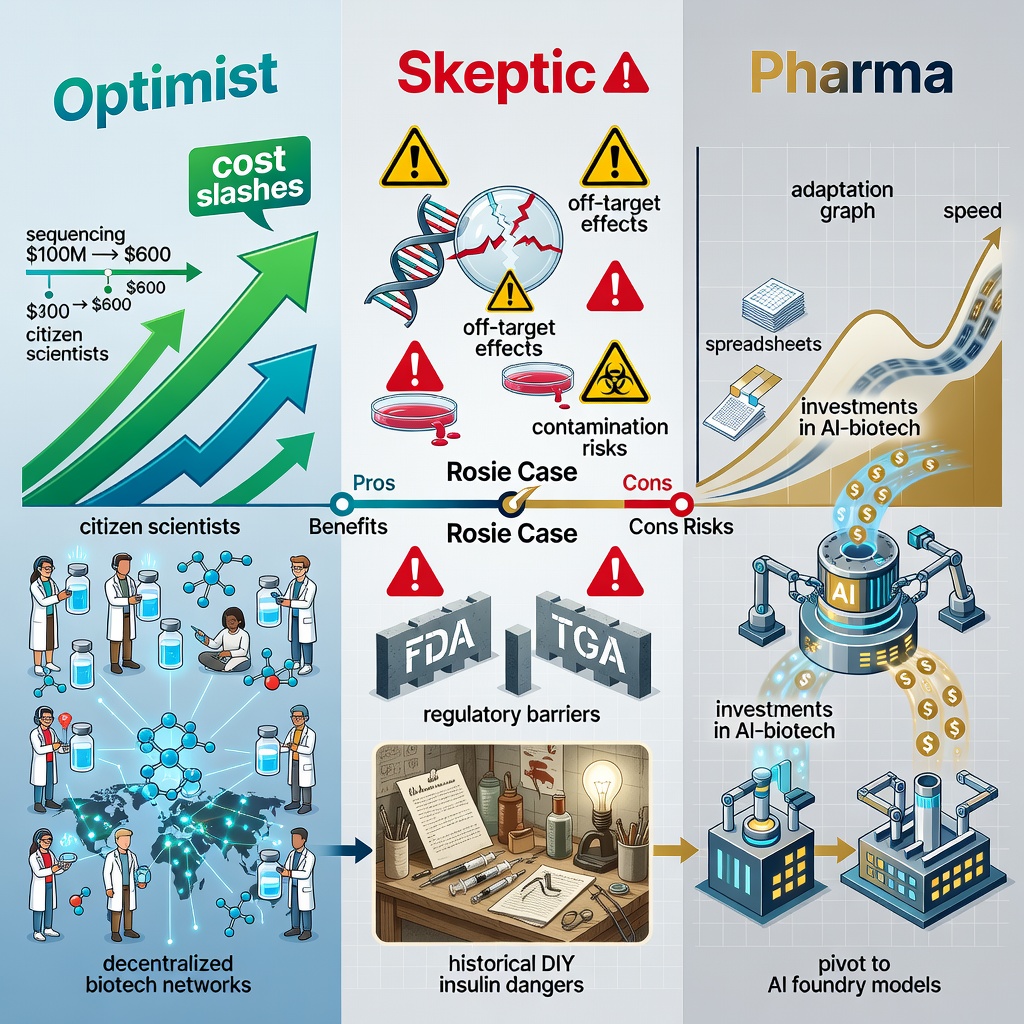
The Optimist’s View: Liberation from Pharma Gatekeepers
Big Pharma’s model favors profits over patients. A 2023 Deloitte report notes 90% of pipeline drugs fail, yet executives greenlight only those projecting $1B+ annual sales. AI sidesteps this: open tools like AlphaFold3 (2024 upgrade) and emerging platforms (e.g., Hugging Face’s bio-models) empower “citizen scientists.” Conyngham’s success validates decentralized biotech parents hacking rare pediatric cancers, communities pooling for orphan drugs. Economically, it slashes costs: sequencing dropped 100,000-fold since 2003 (per NIH), from $100M to $600 per genome.
The Skeptic’s Caution: Risks of Unregulated DIY Medicine
Critics, including FDA regulators, warn of dangers. Veterinary use evades human trial rigor; what if off-target effects emerge? Rosie’s case lacks peer-reviewed publication (as of now), raising reproducibility questions. Bioethicists like Arthur Caplan fret “garage gene therapy” could spawn errors mutagenic vaccines or antibiotic resistance. Legally, Australia’s TGA permits vet experimentation, but human parallels invite scrutiny. Historical parallels? The 1980s DIY insulin movement by diabetics bypassed pharma but risked contamination.
Pharma’s Stance: Adaptation or Obsolescence?
Executives dismiss it as “one-off,” but insiders whisper disruption. Novartis and Roche invest billions in AI-biotech arms. Yet Conyngham exposes a truth: many therapies die not from science, but spreadsheets. AI’s speed (weeks vs. years) forces reinvention pharma may pivot to “AI foundry” models, manufacturing citizen designs for fees.
Future Impacts: Speculating a Biotech Renaissance
Rosie’s story foreshadows seismic shifts by 2030:
– Personalized Medicine Explosion: With sequencing at $100/genome and AI like OpenFold (open-source AlphaFold rivals), weekend warriors could design mRNA for glioblastomas or ALS. Human trials (e.g., BioNTech’s individuated vaccines) accelerate via citizen data-sharing on platforms like PatientsLikeMe.
– Community Biohubs: Makerspaces evolve into “BioFabs” 3D printers for lipids, sequencers on demand. Imagine Etsy for vaccines: upload your genome, get a custom shot.
– Economic Tsunami: Pharma’s $1.5T market fragments; startups like Roswell Biotechnologies (AI proteins) boom. Jobs shift from wet-lab drudgery to prompt engineering.
– Global Equity: In low-resource nations, AI bridges gaps skipping billion-dollar labs for phone-based sequencing apps.
Speculative downsides? Regulatory whack-a-mole: FDA may mandate “AI Rx” certification, stifling innovation. Black markets for rogue therapies? Possible, akin to dark-web drugs. Ethically, IP battles loom DeepMind’s tools are free, but synthesized vaccines aren’t.
Yet optimistically, this mirrors the internet’s 1990s democratization: from ARPANET to TikTok. Cancer mortality, stagnant at 30% for decades (per WHO), could plummet as bespoke cures proliferate.
| Historical Tech Democratization | Then (Elite Era) | Now (Mass Access) | Medical Parallel (Rosie Effect) | ||
|---|---|---|---|---|---|
| Computing | 1960s mainframes ($millions) | Smartphones ($500) | AI protein modeling (free) | ||
| 3D Printing | Industrial ($100K+) | Desktop ($200) | mRNA synthesis (weeks, $thousands) | ||
| Genomics | Human Genome Project (2003, $3B) | $600 per genome | Pet tumor sequencing ($3K) |
Legacy: From One Dog to a Paradigm Shift
Paul Conyngham didn’t just save Rosie he ignited a fuse. Demis Hassabis publicly lauded the effort, tweeting it exemplified AI’s “democratizing force.” As tools proliferate (Grok’s bio plugins, xAI’s models), the gatekeepers tremble. The next cure won’t hail from Pfizer’s C-suite; it’ll emerge from a bedroom, prompted by love and code.
In refusing “impossible,” Conyngham heralds an era where medicine belongs to the people. Rosie’s joyful leaps are the first bound of a revolution personalized, relentless, and unstoppable.